New paper in ACS Chemical Neuroscience
18 03 2026
Category: Publications
Check out our new paper in ACS Chemical Neuroscience, co-authored by Michał Żebrowski, MSc Eng., and Prof. Grzegorz Litwinienko, in collaboration with researchers from the University of Lodz, the Military Institute of Medicine – National Research Institute (Poland), The University of British Columbia (Canada), and Oklahoma State University (USA).
Structure-Dependent Antioxidant Activity of Ibogalogs: Impact of Methoxy Group Position on the Protective Activity in Model and Synaptosomal Lipid Membranes
ACS Chem. Neurosci. 2026, DOI: 10.1021/acschemneuro.6c00087
Congratulations!
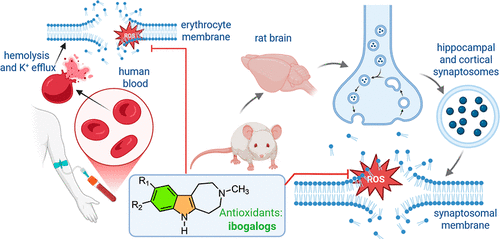
Indole-tetrahydroazepine derivatives of iboga alkaloids, commonly known as “ibogalogs,” are recognized for their antidepressant, anxiolytic, and promnesic effects, primarily mediated through serotonergic targets. However, their role in redox biology and membrane stability has remained largely unexplored. This study provides the first comprehensive physicochemical characterization of three key ibogalogs—ibogaminalog (DM506), ibogainalog (IBG), and tabernanthalog (TBG)—assessing their antioxidant potential and interactions with biological membranes.
Using a multidisciplinary approach, we investigated these compounds in human erythrocytes, rat hippocampal and cortical synaptosomes, and model lipid membranes (liposomes) under AAPH-induced oxidative stress. Our results demonstrate that all three ibogalogs effectively protect human erythrocytes by significantly reducing hemolysis, potassium efflux, and malondialdehyde (MDA) levels—a key marker of lipid peroxidation. Notably, at concentrations ranging from 0.01 to 10 μM, none of the compounds induced membrane damage, confirming a favorable safety profile.
Among the tested derivatives, Tabernanthalog (TBG) exhibited the most robust antioxidant performance, particularly in liposomal systems subjected to lipid-peroxyl-radical-induced peroxidation. In contrast, DM506 and IBG primarily functioned by slowing the rate of autoxidation. These experimental findings were supported by quantum chemical calculations, which indicated that the position and nature of indole substituents—specifically the methoxy group—critically influence bond dissociation enthalpies (BDE), ionization potentials, and radical delocalization.
The study suggests a dual mechanism of action: ibogalogs not only modulate 5-HT transmission but also exert direct neuroprotective effects by scavenging radicals and stabilizing lipid bilayers. Their lipophilic nature likely facilitates integration into biological membranes, reducing vulnerability to peroxidative damage. These findings enhance our understanding of the pharmacological profile of ibogalogs, highlighting their potential as multifunctional agents for treating neurological disorders where oxidative stress plays a pivotal role. Further in vivo studies are warranted to confirm the translatability of these membrane-protective effects in complex biological systems.